Per-patient incidence of selected adverse reactions in adults (Study 4)1
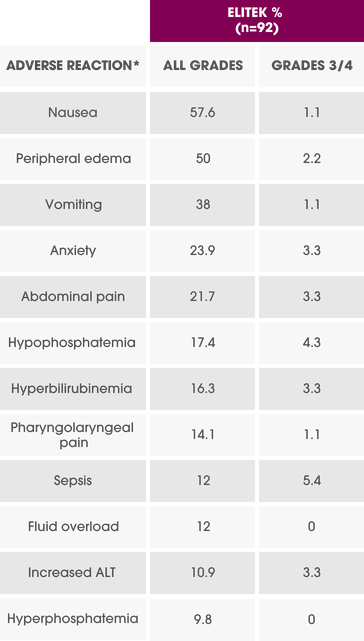
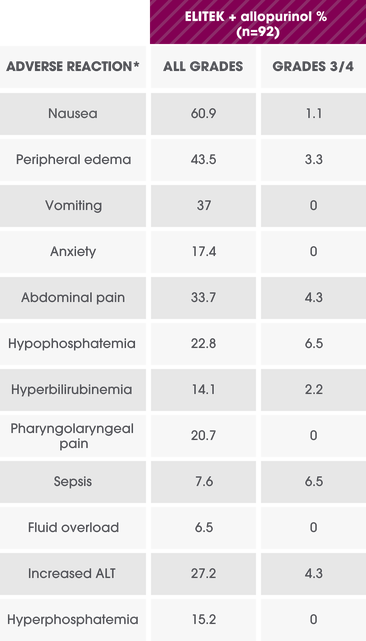
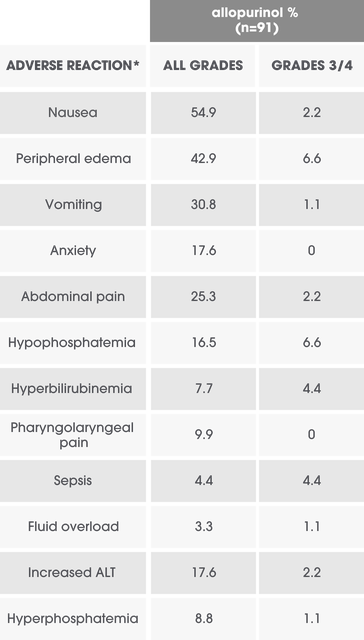
Overall incidence of adverse reactions ≥10% in any ELITEK arm and the difference between any ELITEK arm vs the allopurinol arm ≥5%.1
*Events were reported and graded according to NCI-CTC Version 3.0 and presented as preferred terms MedDRA version 10.1.1
ALT=alanine aminotransferase; NCI-CTC=National Cancer Institute-Common Terminology Criteria.
- Hypersensitivity reactions occurred in 4.3% of ELITEK-treated patients and 1.1% of ELITEK/allopurinol-treated patients in Study 4. Clinical manifestations of hypersensitivity included arthralgia, injection site irritation, peripheral edema, and rash1
- The following serious adverse reactions occurred at a difference in incidence of ≥2% in patients receiving ELITEK compared to patients receiving allopurinol in randomized studies (Study 1 and Study 4): pulmonary hemorrhage, respiratory failure, supraventricular arrhythmias, ischemic coronary artery disorders, and abdominal and gastrointestinal infections1
- The incidence of anaphylaxis, hemolysis, and methemoglobinemia was less than 1% of the 887 ELITEK-treated patients entered on these clinical trials1
Clinician's Guide
Read the Clinician's Guide for a summary of the treatment and patient management of ELITEK
DOWNLOAD NOW
Reference: 1. ELITEK [prescribing information]. Bridgewater, NJ: sanofi-aventis U.S. LLC.

