Pooled efficacy data from 3 studies
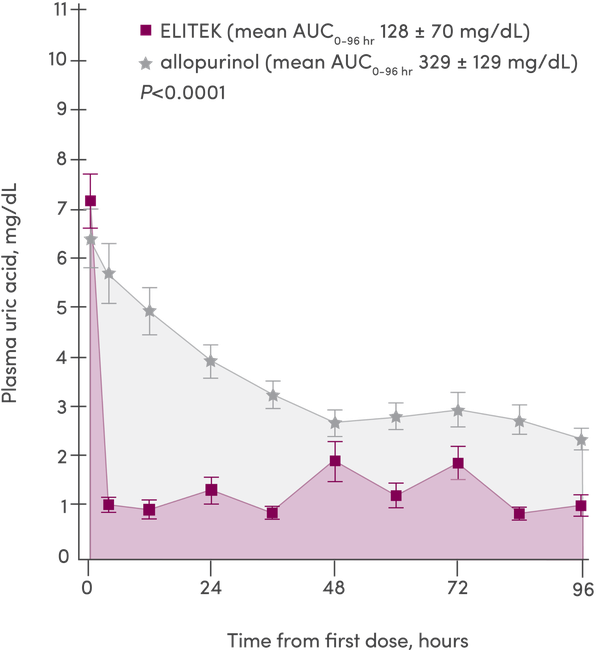
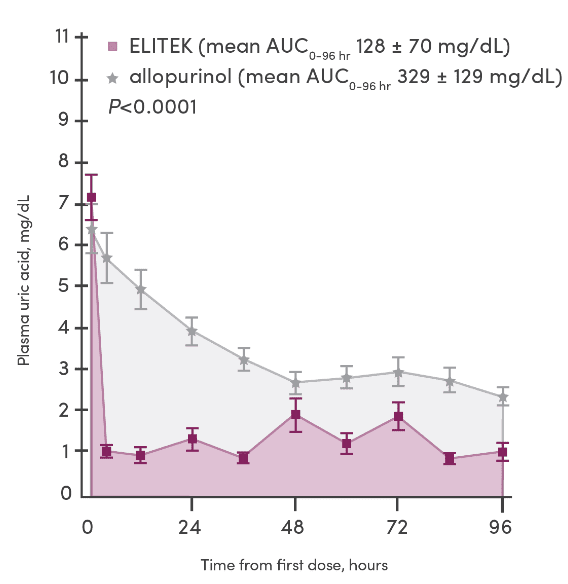
- Data from Study 1, 2, and 3 (n=265) were pooled and analyzed according to the plasma uric acid levels over time1
- 93% (246/265) of patients (pooled) enrolled in these clinical trials were pediatric1
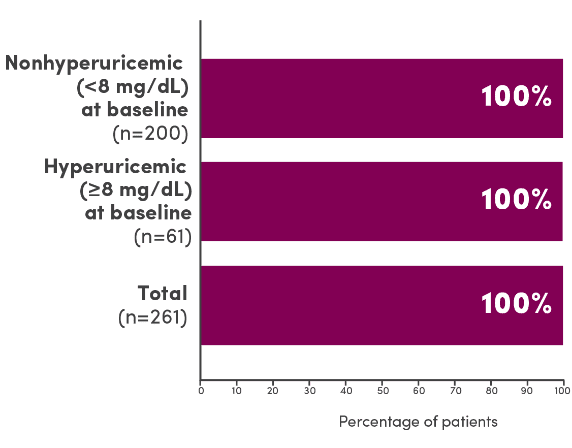
- 98% (261/265) of enrolled patients were evaluable: 77% (200/261) of patients were nonhyperuricemic (<8 mg/dL) at baseline and 23% (61/261) of patients were hyperuricemic (≤8 mg/dL) at baseline1
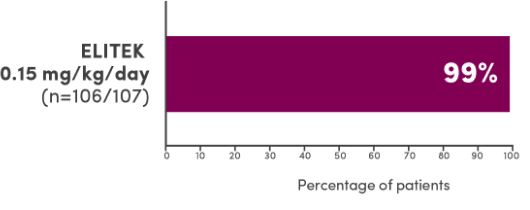
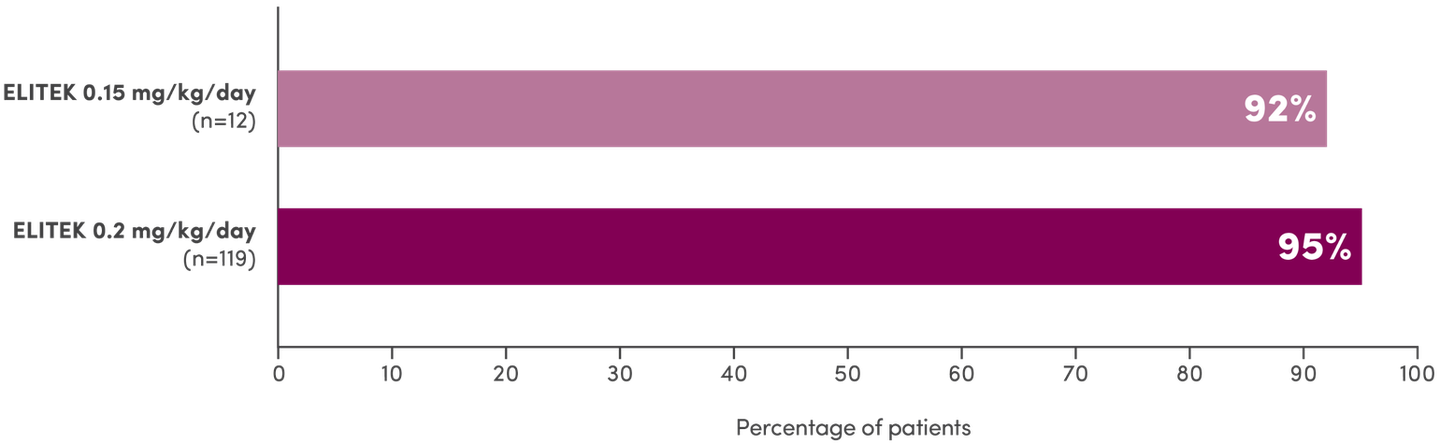
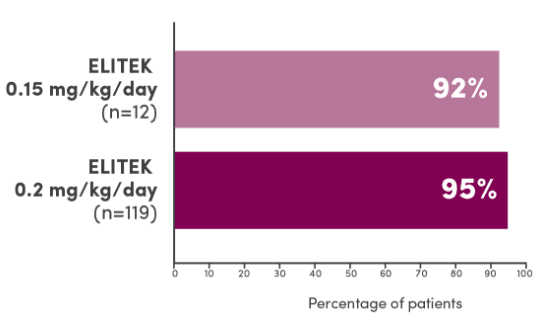
In 3 clinical trials, ELITEK maintained normal uric acid levels in 100% of evaluable patients by Day 41
Evaluable patients maintaining normal uric acid levels (≤6.5 mg/dL for patients <13 years of age or ≤7.5 mg/dL for patients ≥13 years of age) 4 days post-initiation of ELITEK1
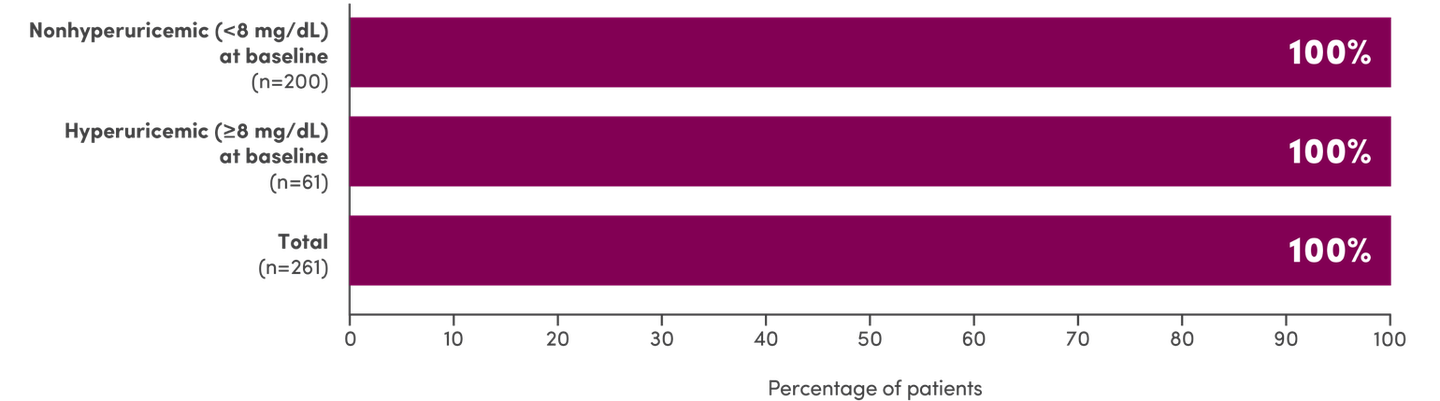
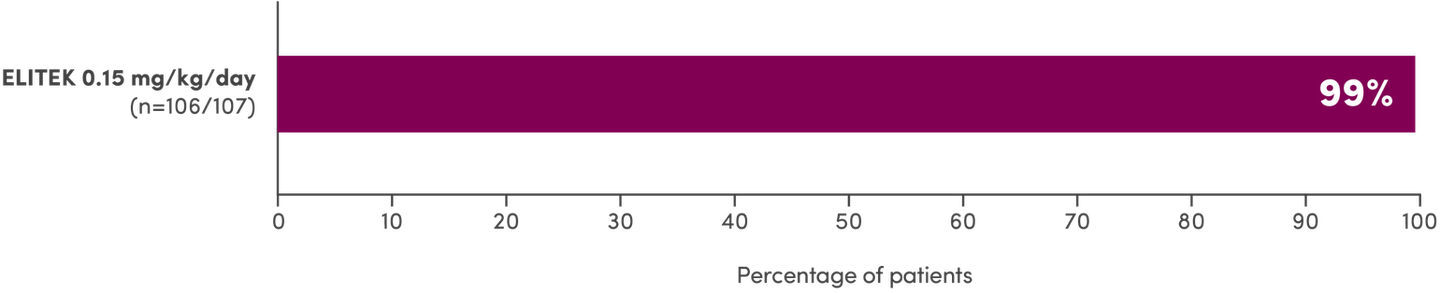
- Of the 261 evaluable patients in Study 1, 2, and 3 (pooled)1:
- Normal uric acid levels were maintained by 92% (240/261) of patients after 4 hours, 93% (245/261) after 24 hours, 97% (254/261) after 48 hours, and 99% (260/261) after 72 hours
- For patients with hyperuricemia (≥8 mg/dL) at baseline (61/261), uric acid levels were maintained by 72% (44/61) after 4 hours, 80% (49/61) after 24 hours, 92% (56/61) after 48 hours, and 98% (60/61) after 72 hours
- Maintenance was defined as uric acid levels ≤6.5 mg/dL (patients <13 years of age) or ≤7.5 mg/dL (patients ≥13 years of age) without the need for allopurinol or other agents1
References: 1. ELITEK [prescribing information]. Bridgewater, NJ: sanofi-aventis U.S. LLC. 2. Goldman SC, Holcenberg JS, Finklestein JZ, et al. A randomized comparison between rasburicase and allopurinol in children with lymphoma or leukemia at high risk for tumor lysis. Blood. 2001;97:2998-3003.